|
In aqueous solution, AgNO 3 + NaCl = AgCl + NaNO 3 - AgCl + NaNO 3 (aqueous) The following are some additional examples of chemical equations for the precipitation reaction: The following is the chemical equation for this reaction:ĢKOH(aqueous)+ CaCl 2 -Aqueous Ca(OH) 2 + 2KCl 2KOH + CaCl 2 (aqueous) The reaction between calcium chloride and potassium hydroxide, which results in the formation of an insoluble salt known as calcium hydroxide, is another example of a precipitation reaction. It is possible to determine the presence of different ions in a particular solution using precipitation reactions, which are useful in many situations. Silver chloride is a solid compound that is insoluble in water. In the preceding reaction, a white precipitate known as silver chloride (AgCl) is formed, which is in the solid-state state. The chemical equation for this precipitation reaction is shown in the following section.ĪgNO 3 (aqueous) + KCl(aqueous) -– AgCl(precipitate) + KNO 3 (aqueous) An example of an insoluble salt that results from the precipitation reaction is shown in the diagram below. In the chemical reaction between potassium chloride and silver nitrate, solid silver chloride is precipitated out as a byproduct, which is one of the most well-known examples of precipitation reactions. The formation of insoluble salts that precipitate out of solution can also occur when two or more solutions containing different salts are combined. When a precipitation reaction occurs, it is typically a double displacement reaction that results in the formation of a solid form residue known as the precipitate. One of these products is insoluble in the solution and must be precipitated out (and is referred to as the ‘precipitate’ in this context).Īccording to the definition, a precipitation reaction is “a chemical reaction that occurs in an aqueous solution in which two ionic bonds combine, resulting in the formation of an insoluble salt.” Precipitates are the insoluble salts that form during precipitation reactions and are formed during the process of precipitation. During such reactions, two different soluble salts (both of which are present in aqueous solutions) react with one another to form two products. Precipitation reactions are a type of chemical reaction that is commonly encountered. Burning, corrosion, cooking food, and digestion are all examples of such reactions that occur frequently. There are numerous chemical reactions that take place in our daily lives. The reactants can exist in any phase, including solid, gaseous, and liquid. 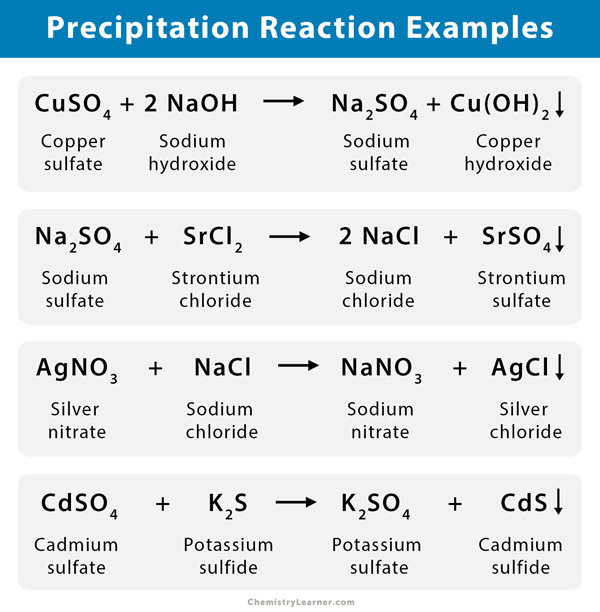
These are the compounds that take part in a chemical reaction and are referred to as reactants. These reactions are represented by chemical equations, which show both the reactants and the products involved in the reactions. We can better understand chemical reactions between different elements or compounds if we use chemical equations to aid in our understanding. Chemical reactions are characterised by chemical changes that result in the formation of new compounds when certain conditions are met.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
